Press-room / news / Science news /
TLR2 and Zinc: A Surprising Partnership in Immune Signaling
A team of scientists from the Laboratory of Biomolecular NMR Spectroscopy at the Institute of Bioorganic Chemistry of the Russian Academy of Sciences, in collaboration with researchers from China, has made a breakthrough in understanding the function of Toll-like receptor 2 (TLR2), a key component of the innate immune system. Their study, published in FEBS Letters, reveals that TLR2 exhibits a previously unknown ability to bind zinc ions with high affinity and specificity. Toll-like receptors (TLRs) serve as the first line of defense in the immune system by recognizing pathogens and initiating immune responses. Despite extensive research, the precise molecular mechanisms governing TLR activation remain elusive. The newly identified zinc-binding capability of TLR2 provides fresh insights into its regulatory mechanisms. The researchers also identified specific amino acids essential for zinc coordination and TLR2 function, highlighting a potential link between zinc homeostasis and immune activation. These findings suggest that zinc plays a critical role in modulating TLR-mediated immune signaling, opening new avenues for research into immune system regulation and potential therapeutic applications.
Our immune system relies on Toll-like receptors (TLRs) to recognize invaders and trigger a defense response. But how exactly do these receptors work? Scientists have long been puzzled by the molecular mechanisms behind their activation. A recent study, published in FEBS Letters, sheds new light on this mystery—revealing that zinc ions play a crucial role in TLR function.
Previously, researchers discovered that TLR1, a key immune receptor, binds zinc ions with remarkable strength. This interaction is controlled by two specific cysteine amino acids, one of which is essential for the receptor’s activation. Since TLR1 partners with another receptor, TLR2, to perform its immune duties, the scientists wondered: Could TLR2 also interact with zinc?
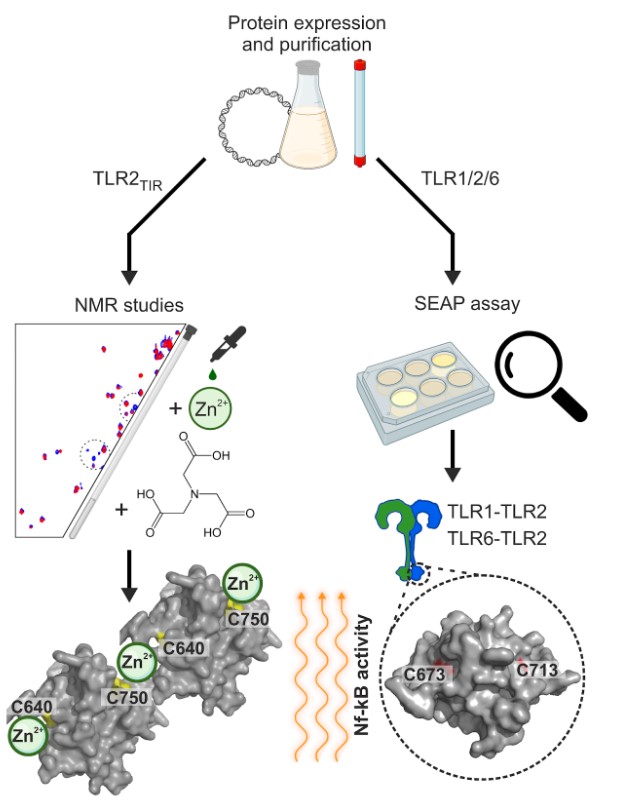
To find out, the team conducted a series of experiments. First, they showed that TLR2 competes with TLR1 for zinc, suggesting that both receptors have a similar ability to bind this metal. But there was an unexpected twist—unlike what was previously believed, TLR1 and TLR2 did not interact directly in these conditions. Instead, when TLR2 bound zinc, it formed large complexes with other TLR2 molecules. This strong binding meant the researchers had to develop a special technique to measure the stability of a protein/metal complex. Using a chelating agent called NTA, they determined that the TLR2-zinc complex was extremely stable, with a dissociation constant of just 6 nM.
Digging deeper, the scientists used genetic engineering techniques to alter specific amino acids in TLR2. Since cysteine is commonly found in proteins that bind zinc, they mutated these residues to see how it affected the receptor’s ability to interact with the metal. The results were striking—mutating all cysteines nearly eliminated the receptor’s ability to bind zinc. When they altered only one or two at a time, no single cysteine emerged as the most important; instead, all were necessary for the receptor’s ability to form zinc-mediated complexes.
Finally, colleagues from the Changchun Institute of Applied Chemistry (China) revealed that two of TLR2’s cysteines are particularly important for its role in the immune response. Mutating these residues prevented the activation of the receptor, suggesting that zinc may play a direct role in triggering immune signaling pathways.
These findings challenge previous assumptions about how TLRs function and highlight zinc as a key player in the immune system. Supported by grant No. 22-14-00020 from the Russian Science Foundation, this study opens up new questions about how metals like zinc contribute to immune system regulation—and whether targeting these interactions could lead to new treatments for immune-related diseases.
march 31

